Tissue cross-reactivity (TCR) studies are key in the development of monoclonal antibodies and other related biologics to ensure that the experimental antibody or biologic does not bind to epitopes other than the target site as this could lead to treatment-related toxicity. TCR studies are also useful in identifying target species for toxicology studies as well as target organs.
Comparative Biosciences, Inc’s. TCR services support the development of your candidate therapeutic antibodies by providing a powerful immunohistochemical assessment of potential cross-reactivity with a range of animal tissue. CBI has all the organizations defined in the relevant FDA and EMA submissions in accordance with the FDA’s recommendation and is in full compliance with local legal regulations and ethical norms.
Research Tissues Applications
- Complete tissue bank of a variety of species including transgenic/knockout models, diseased tissue and tumors; frozen and fixed tissues for human tissue cross-reactivity studies; and in vivo studies
- Comprehensive, high-quality audited reports
- Digital photomicroscopy
- Digital image analysis and histomorphometry
- Quality assurance with critical phase audits
- Rapid scheduling and reporting
- Reasonable cost-effective pricing
- Strong expertise in immunohistochemistry, histology and immunopathology with both animal and human tissues
- Study-specific protocols prepared to reflect your exact requirements
- Tissue evaluation by experienced ACVP board-certified veterinary pathologist
- Tumor bank contains a wide variety of common, rare and unusual human tumors
CBI Shows How Preclinical Studies Can Support IND Applications and CTA’s
It is extremely costly to have a therapeutic antibody or antibody-like molecule fail during clinical development. Therefore, drug developers need to mitigate any potential issues before first-in-human studies begin. Many preclinical investigations that are essential components of a regulatory Investigational New Drug (IND) application or Clinical Trial Application (CTA) are intended to minimize the risk of harm when a drug is first given to humans. One such preclinical safety study is the assessment of tissue cross-reactivity (TCR)
Our studies are conducted in three phases:
Phase 1: Initial Characterization and Optimization
For the initial characterization and optimization, the optimal specificity and staining conditions for the experimental antibody is determined.
Phase 2: Preliminary Tissue Cross-Reactivity Screening
Using the optimal specificity and staining conditions, a preliminary study with the standard 37 tissues is conducted and evaluated, including:
- Adrenal gland
- Bladder
- Blood cells
- Bone marrow
- Breast
- Cerebellum
- Cerebral cortex
- Colon
- Endothelium
- Eye
- Fallopian tube
- Gastrointestinal tract – colon, esophagus, small intestine, stomach
- Heart
- Kidney
- Liver
- Lung
- Lymph node
- Ovary
- Pancreas
- Parathyroid
- Peripheral nerve
- Pituitary
- Placenta
- Prostate
- Salivary gland
- Skeletal muscle
- Skin
- Spinal cord
- Spleen
- Testes
- Thymus
- Thyroid
- Tonsil
- Ureter
- Uterus
Phase 3: Full Cross-Reactivity Study
Following optimization in Phase 1 and 2, a full cross-reactivity study can be performed in accordance with the FDA’s recommendation.
In these studies:
- Tissues used from three unrelated adults
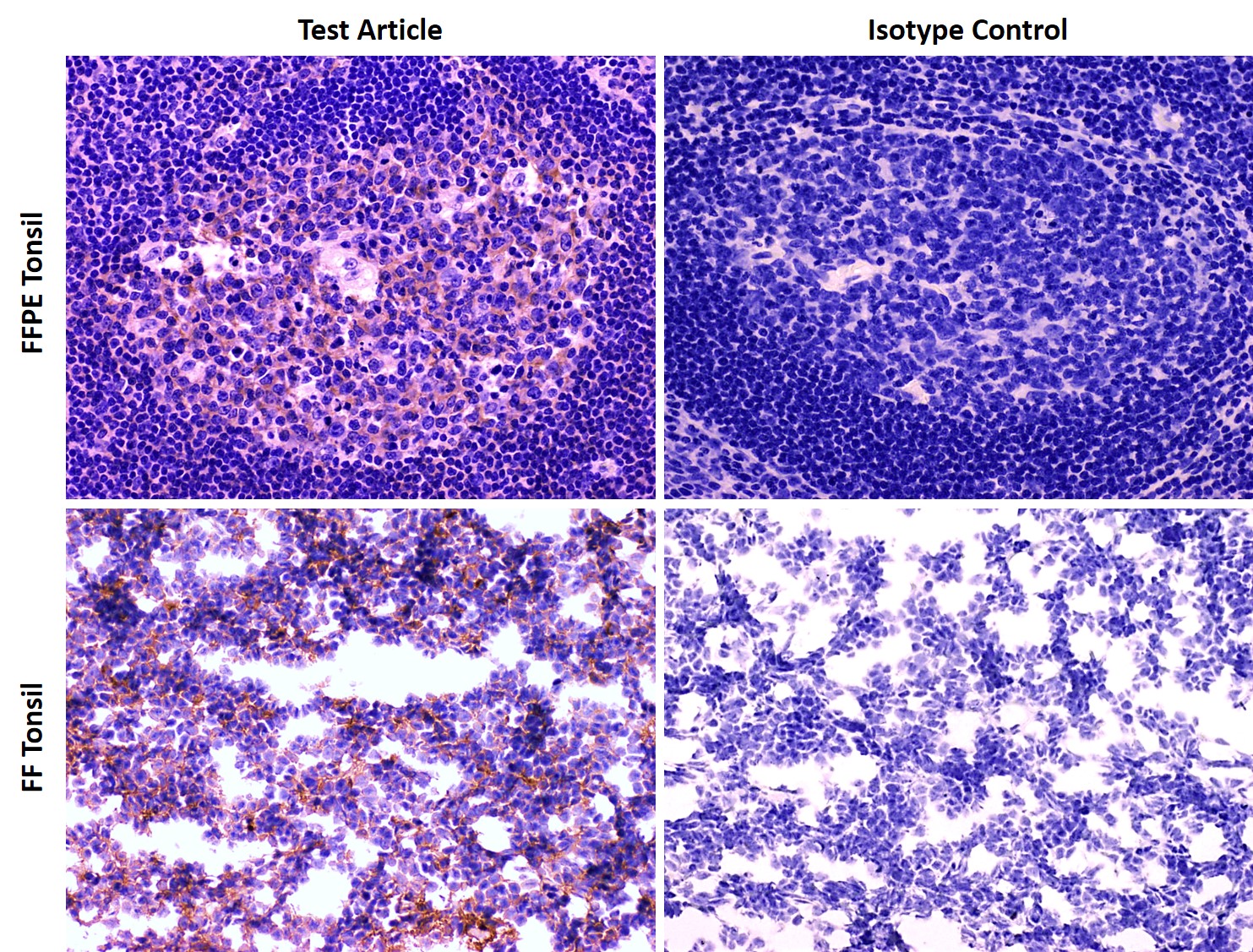
- Experimental antibody at two dose levels
- Isotype control antibody
- β2 microglobulin on each tissue for staining control
- Evaluation by ACVP board-certified veterinary pathologist
- Staining intensity scored 0-4
- Conducted under GLP conditions with audits of critical phases
- Final report suitable for FDA submission
- Archiving
CBI Shows How Preclinical Studies Can Support IND Applications and CTA’s
It is extremely costly to have a therapeutic antibody or antibody-like molecule fail during clinical development. Therefore, drug developers need to mitigate any potential issues before first-in-human studies begin. Many preclinical investigations that are essential components of a regulatory Investigational New Drug (IND) application or Clinical Trial Application (CTA) are intended to minimize the risk of harm when a drug is first given to humans. One such preclinical safety study is the assessment of tissue cross-reactivity (TCR)

Contact Comparative Biosciences, Inc. to discuss a scientific study program for Tissue Cross – Reactivity Studies and Services
Comparative Biosciences, Inc. · Jonathan A. Geller, MS · Business Development Manager · Phone: 408.738.9260


